U.S. Department of Energy’s Argonne National Laboratory researchers have made groundbreaking innovations. They created a fluoride electrolyte that could safeguard the performance of a future battery, preventing its decline over time.
Sodium fluoride, a compound containing fluorine, is commonly found in toothpaste as it helps safeguard teeth from decay. However, scientists have developed its substance for future needs.
Zhengcheng Zhang, a group leader in Argonne’s Chemical Sciences and Engineering division, explained that on-lithium-ion batteries possess chemical compositions that allow storing two or more times the amount of energy per volume or weight compared to lithium-ion batteries. This increased energy storage capacity can significantly enhance the driving range of vehicles, and there is even a possibility of powering long-haul trucks and aircraft in the future.
“The utilisation of our fluorinated cation electrolyte in lithium metal batteries has the potential to enhance the electric vehicle sector. Moreover, the applicability of this electrolyte extends beyond lithium-ion batteries to various other advanced battery systems,” Zhang said.
The widespread adoption of these batteries is anticipated to contribute to mitigating the challenges of climate change. However, a key drawback is that their high energy density diminishes rapidly with repeated charging and discharging cycles.
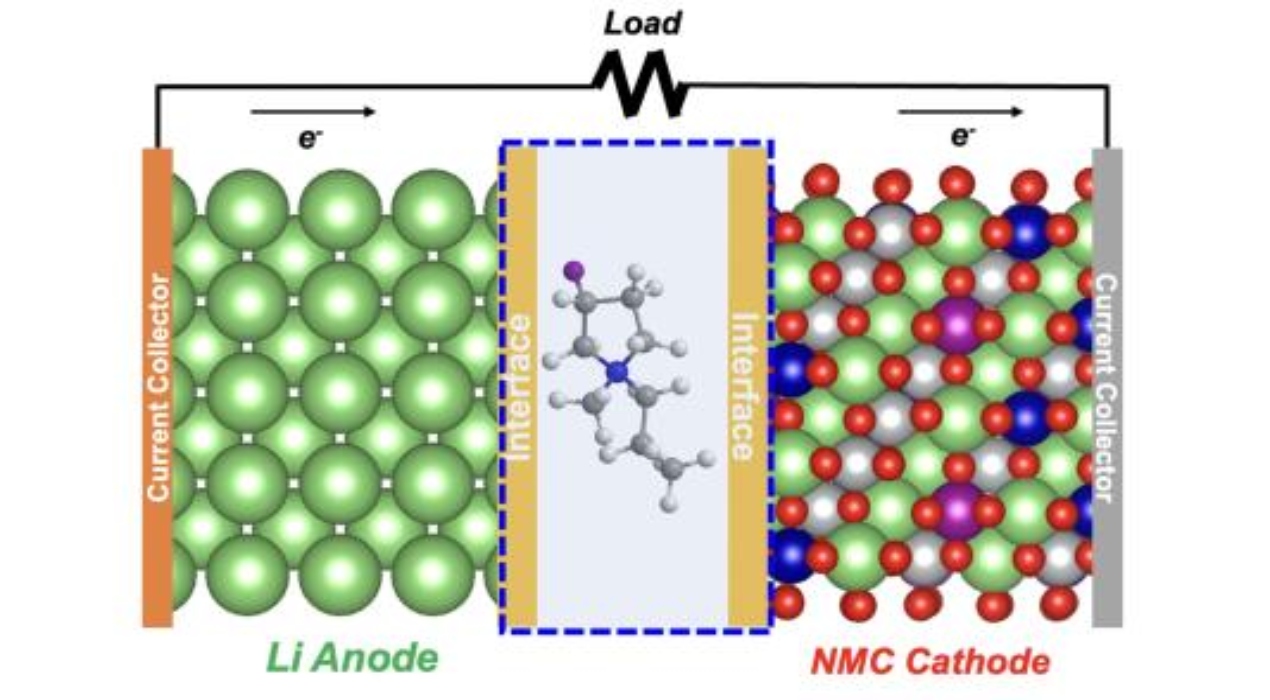
One of the leading candidates in battery technology features a lithium metal anode instead of the typical graphite found in lithium-ion batteries, earning it the name “lithium metal” battery. On the other hand, the cathode is composed of a metal oxide containing nickel, manganese, and cobalt (NMC). While this design enables a significantly higher energy density than lithium-ion batteries, this exceptional performance diminishes rapidly after only a few dozen charge-discharge cycles.
The team’s solution involved modifying the electrolyte. This liquid facilitates the movement of lithium ions between the cathode and anode during the charging and discharging process in lithium metal batteries. Typically, this electrolyte comprises a lithium-containing salt dissolved in a solvent.
The issue leading to the temporary cycle-life problem lies in the electrolyte’s inability to form a sufficient protective layer on the anode’s surface during the initial cycles. This protective layer, known as the solid-electrolyte-interphase (SEI), serves as a barrier, allowing lithium ions to move freely in and out of the anode during battery charging and discharging.
The research team identified a novel fluoride-based solvent that effectively sustains a durable protective layer for numerous cycles. This achievement involves combining a positively charged fluorinated component (cation) with a distinct negatively charged fluorinated component (anion), resulting in what scientists refer to as an ionic liquid. An ionic liquid is a liquid composed of positively and negatively charged ions.
“The primary distinction in their novel electrolyte lies in replacing hydrogen atoms with fluorine atoms within the ring-shaped structure of the cation component of the ionic liquid. This specific alteration proved to be crucial in preserving optimal performance over hundreds of cycles in a test lithium metal cell,” Zhang emphasised.
By adjusting the fluoride solvent to lithium salt ratio, the research team successfully engineered a layer with optimal characteristics, including an ideal thickness for the solid-electrolyte-interphase (SEI). This well-calibrated layer facilitated the efficient flow of lithium ions between the electrodes during the charging and discharging processes, sustaining this performance over numerous cycles.
Furthermore, the team’s innovative electrolyte offers several additional benefits. It boasts a low cost due to its ability to produce high purity and yield through a single, straightforward step, eliminating the need for multiple processes. The electrolyte is also environmentally friendly as it utilises a reduced amount of solvent, which is volatile and can potentially release contaminants into the environment. Additionally, the new electrolyte enhances safety measures as it is non-flammable.
Zhang expressed confidence in the substantial impact that lithium metal batteries equipped with the fluorinated cation electrolyte could have on the electric vehicle industry. Furthermore, the versatile nature of this electrolyte undoubtedly extends its usefulness to advanced battery systems beyond the scope of lithium-ion, presenting promising opportunities for various applications in the field.
















