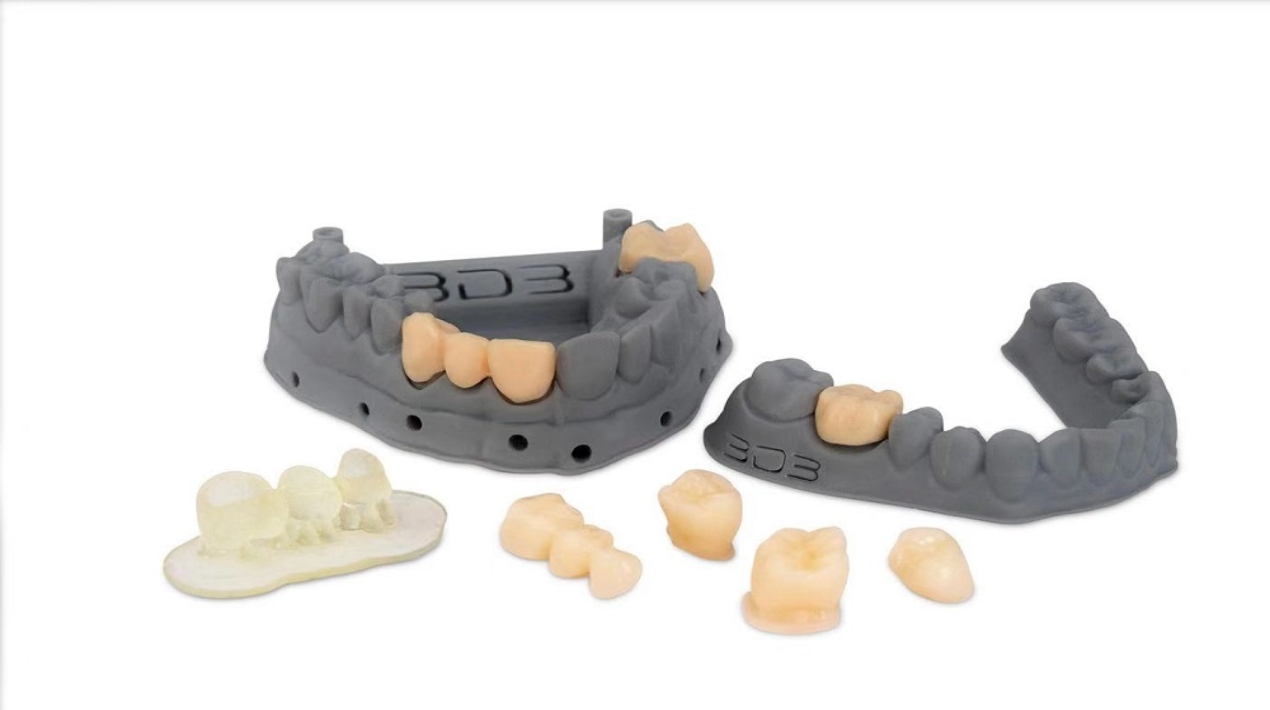
Using a desk-side 3D printer, dentists can now build a permanent crown material in just 15 minutes while also accurately replicating the patient’s natural tooth colour without the use of additional colouring. This solution saves dentists and patients a significant amount of time when compared to the weeks the process would have previously taken.
A*STAR’s Institute of Materials Research and Engineering (IMRE) contributed technical expertise and experience to the project, creating a specially formulated resin with excellent strength, rigidity, and biocompatibility that mimics tooth enamel.
The agency has licenced a method for producing a three-dimensional object layer-by-layer using a computer-generated design, thereby accelerating the research and development period, and accelerating the commercialisation of the material.
A dental crown is one of the most common dental procedures used to restore or repair teeth, but it is still a time-consuming procedure. Singapore is attempting to shorten appointment times by using innovative 3D-printed dental crowns.
Patients must visit the dentist at least twice when getting a dental crown. The first visit consists of preparing the tooth and taking an impression of the permanent crown’s construction. The impression is then sent to a third-party dental laboratory to be made into a crown. This procedure can take two to three weeks, which is inconvenient for the patient. Multiple appointments also take up dentists’ time, which could be better spent treating more patients.
As a result, it is not surprising that the dental industry sees 3D-printed dental crowns as a viable solution. Instead of waiting for the laboratory, dentists could print the crown in their clinic, and patients could be fitted with their new crown in a single appointment.
Although 3D printing technology is mature, creating a material that meets healthcare regulatory standards remains difficult. The material must also be strong enough to withstand wear and tear while remaining biocompatible.
The solution consisted of two parts: first, 3D printing technology, and second, developing the right material to print the crowns with. As it is intended to last for several years, the crown material must match actual tooth enamel strength and have high biocompatibility. It must also meet some regulatory requirements.
It has been difficult to develop a material that meets all the criteria for a permanent dental crown in the market, however, A*STAR intends to develop new and unique dental needs, as well as other dental products. More than 20 customers in Singapore use the technology’s 3D printed dental crown solution, as do international customers in China, Hong Kong, Macau, and Malaysia.
OpenGov recently reported that researchers from the Faculty of Dentistry at the University of Hong Kong (HKU) and the Department of Computer Science of Chu Hai College of Higher Education, collaborated to develop a new approach using artificial intelligence to automate the design of individualised dentures, to enhance the treatment efficiency and improve patient experience.
The AI technology used in the process was based on 3D Generative Adversarial Network (3D-GAN) algorithm and tested on 175 participants recruited at HKU. The study shows that AI technology could reconstruct the shape of a natural healthy tooth and automate the process of false teeth design with high accuracy.
Meanwhile, the Health Sciences Authority (HSA) of Singapore has collaborated with four other international regulatory agencies as part of the Access Consortium to expedite the approval of a new therapeutic product for the treatment of two eye diseases that are among the leading causes of irreversible vision loss.
Through the “New Active Substance Work Sharing Initiative” (NASWSI) of the Access Consortium, a five-way product evaluation was conducted for the first time. This multi-agency programme aims to expedite patients’ access to innovative medications and treatments by improving the regulatory review process and reducing duplication of efforts on the part of both regulators and pharmaceutical companies.
The Access Consortium is a group of like-minded international health product regulators. With the introduction of its newest consortium member, the UK MHRA, in January 2021, it was renamed the previous Australia-Canada Singapore-Switzerland Consortium (ACSS). The new name reflects the group’s primary goal of providing patients in member countries with timely access to high-quality, safe, and effective therapeutic products.















