Scientists from China's National Center for Nanoscience and
Technology (NCNST) have successfully created and tested the world's first
autonomous DNA nanorobots to combat cancer tumours, according to
state media outlet, Xinhua. This was accomplished in collaboration
with researchers from Arizona State University (ASU). A study with successful
demonstration of the technology in mammals, using breast-cancer, melanoma,
ovarian and lung-cancer mouse models, was published in the journal Nature
Biotechnology.
The work was supported by grants from National Basic
Research Plan of China (MoST Program), the National Natural Science Foundation
of China, Innovation Research Group of National Natural Science Foundation,
Beijing Municipal Science & Technology Commission, CAS (Chinese Academy of Sciences)
Interdisciplinary Innovation Team, K. C. Wong Education Foundation and the US
National Institutes of Health Director’s Transformative Research Award.
A nanorobot refers to a system designed to perform a
specific task at nanoscale dimensions. The DNA nanorobots provide a highly
precise and targeted treatment method, destroying only the tumours by cutting
off their blood supply. Surrounding health tissue is not harmed unlike in chemotherapy
and radiation.
This technology provides a strategy that can be used for
many types of cancer, since all solid tumour-feeding blood vessels are
essentially the same, said Hao Yan, director of the ASU Biodesign Institute’s
Center for Molecular Design and Biomimetics and the Milton Glick Professor in
the School of Molecular Sciences.
This has been described as a milestone in the new field of nanomedicine,
which seeks to use nanotechnology to create minuscule, molecule-sized
nanoparticles to diagnose and treat difficult diseases, especially cancer. The
challenge is to design, build and carefully control nanorobots to actively seek
and destroy cancerous tumours, while not harming any healthy cells.
The research was started five years ago, when NCNST
researchers first looked at cutting off blood supply to tumours by inducing
blood coagulation using DNA-based nanocarriers. Through the collaboration with
ASU, the nanomedicine design was upgraded to be a fully programmable robotic
system, able to perform its mission entirely on its own.
For the study the scientists used a well-known mouse tumour
model, where human cancer cells are injected into a mouse to induce aggressive
tumour growth. Nanorobots were deployed once the tumour was growing.
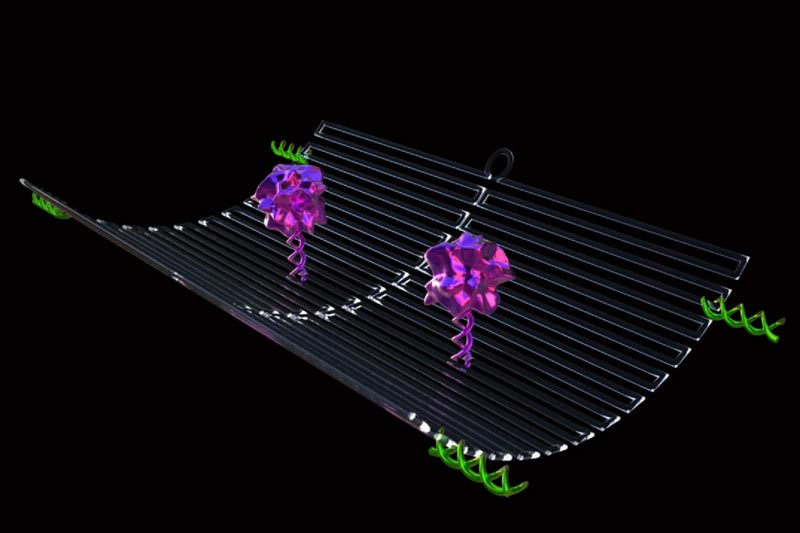
Each nanorobot is made from a flat, rectangular DNA origami
sheet, 90 nanometres by 60 nanometres in size. A key blood-clotting enzyme,
called thrombin, is attached to the surface. Thrombin can block tumour blood
flow by clotting the blood within the vessels that feed tumour growth, leading
to tumour tissue death.
An average of four thrombin molecules was attached to a flat
DNA scaffold and the flat sheet was folded in on itself like a sheet of paper
into a circle to make a hollow tube. They were then injected into a mouse,
where they travelled through the bloodstream looking for the tumours.
To ensure that the nanorobots only attacked cancer cells, a
special payload was included on the surface of the nanorobot, called a DNA
aptamer. The DNA aptamer could specifically target a protein, called nucleolin,
that is made in high amounts only on the surface of tumour endothelial cells and
is not found on the surface of healthy cells.
The nanorobot was programmed to release its drug cargo inside
the tumour and expose the thrombin after binding to the tumour blood vessel
surface.
The nanorobots congregating in large numbers rapidly
surrounding the tumour just hours after injection. The treatment successfully blocked
tumour blood supply and caused tumour tissue damage within 24 hours while
having no effect on healthy tissues. Being a natural biocompatible and
biodegradable material, most of the nanorobots were cleared and degraded from
the body after 24 hours. By two days, there was evidence of advanced thrombosis,
and at the three-day point, thrombi in all tumor vessels were observed.
There was no evidence of the nanorobots spreading into the
brain where they could cause unwanted side effects, such as a stroke.
“The nanorobot proved to be safe and immunologically inert
for use in normal mice and, also in Bama miniature pigs, showing no detectable
changes in normal blood coagulation or cell morphology,” said Yuliang Zhao,
also a professor at NCNST and lead scientist of the international collaborative
team.
There was no evidence of the nanorobots spreading into the
brain where they could cause unwanted side effects, such as a stroke.
In the melanoma mouse model, the median survival time more
than doubled, extending from 20.5 to 45 days. The system was also tested in a
primary mouse lung-cancer model, which mimics the human clinical course of
lung-cancer patients. Shrinkage of tumour tissues was demonstrated after a
two-week treatment.
Nie Guangjun, professor at the NCNST and a key member of the
collaborative team said that they are currently working with a biotech firm to
do pre-clinical studies and hopefully translate the technology into a viable
anti-tumour therapeutic.
















